iHealth Covid-19 Home Test (2)
Tests for Covid-19
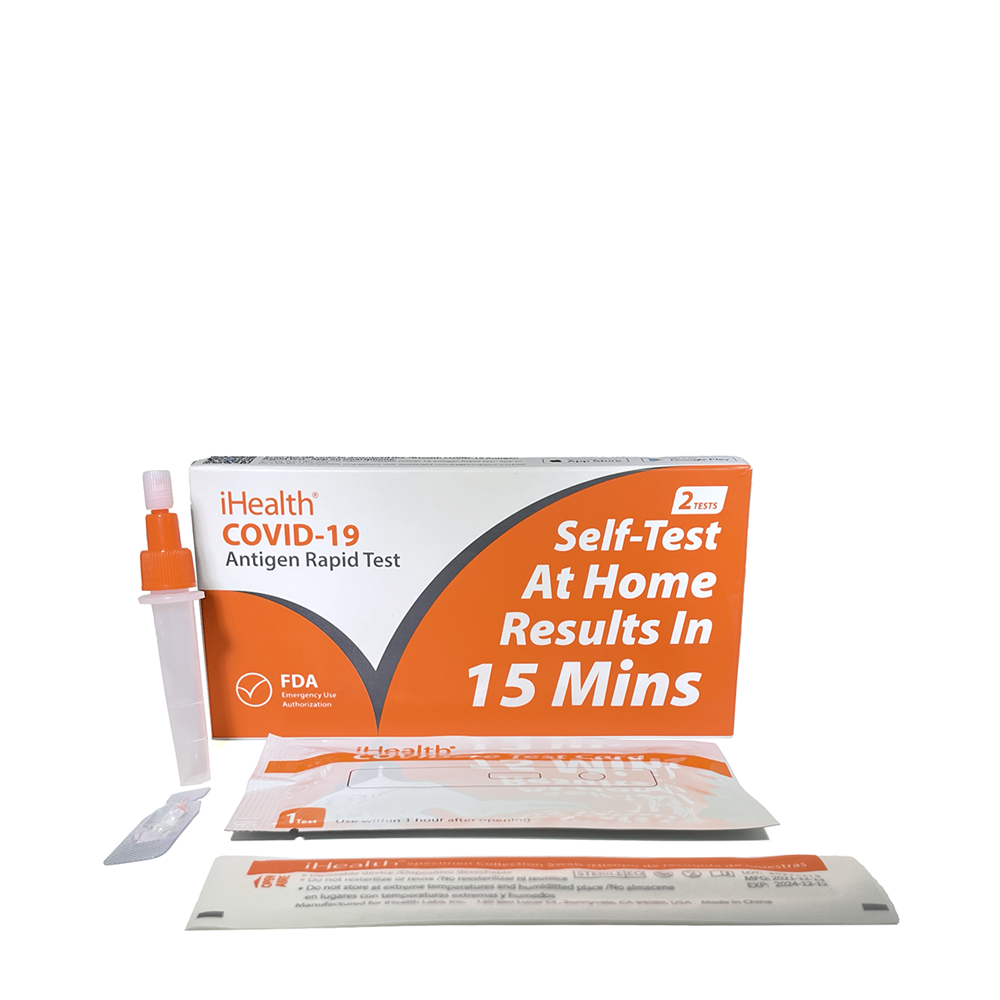
2 tests per kit
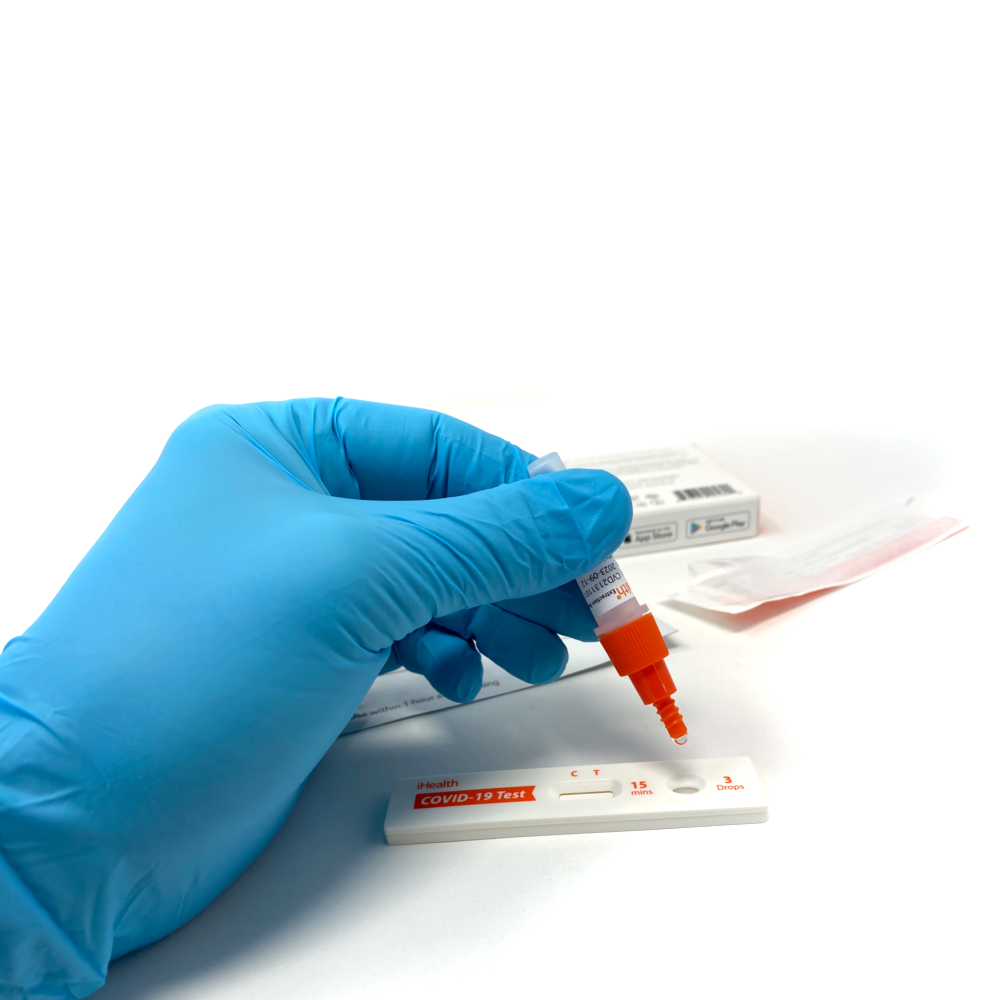
Results in 15 minutes
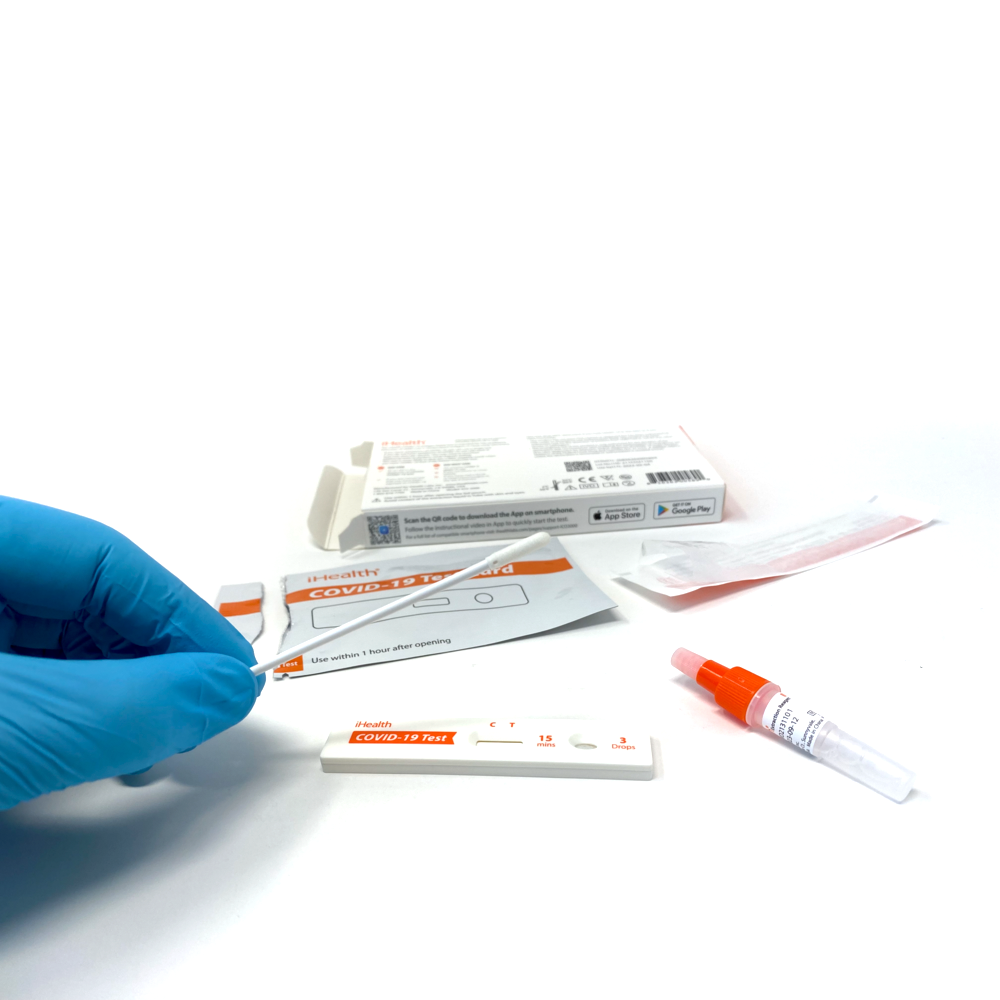
Lower nasal swab
Accuracy: 94.3% PPA, 98.1% NPA
Tests for Covid-19
2 tests per kit
Results in 15 minutes
Lower nasal swab
Accuracy: 94.3% PPA, 98.1% NPA
Tests for Covid-19
2 tests per kit
Results in 15 minutes
Lower nasal swab
Accuracy: 94.3% PPA, 98.1% NPA
Specifications
Home test for Covid-19
Test type: Rapid Antigen
Run time: 15 minutes
Collection type: Lower nasal swab
Covid-19: 94.3% PPA, 98.1% NPA
Manufacturer: iHealth Labs
Storage & Expiration
Store at 2-30°C (35-86°F)
Do not freeze any contents of the kit.
Reagents and materials are stable until expiration date printed on outer packaging.
Test device must remain in the sealed pouch until use.
Box Components
Test devices (2)
Nasal collection swabs (2)
Solution vial (2)
Instructions For Use (1)
Manuals & Documents
Intended Use
The iHealth COVID-19 Antigen Rapid Test is a lateral flow assay intended for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2.
This test is authorized for non-prescription home use with self-collected anterior nasal (nares) swab samples from individuals aged 15 years or older with symptoms of COVID-19 within the first 7 days of symptom onset. This test is also authorized for non-prescription home use with adult-collected anterior nasal (nares) swab samples from individuals aged 2 years or older with symptoms of COVID-19 within the first 7 days of symptom onset.
This test is also authorized for non-prescription home use with self-collected anterior nasal (nares) swab samples from individuals aged 15 years or older, or adult-collected anterior nasal (nares) swab samples from individuals aged 2 years or older, with or without symptoms or other epidemiological reasons to suspect COVID-19 when tested twice over three days with at least 24 hours (and no more than 48 hours) between tests.